
Therapeutic devices require manufacturing processes that go far beyond standard electronics—processes that ensure zero-defect performance under extreme operational conditions. Our manufacturing infrastructure is custom-built to meet this rigorous demand:
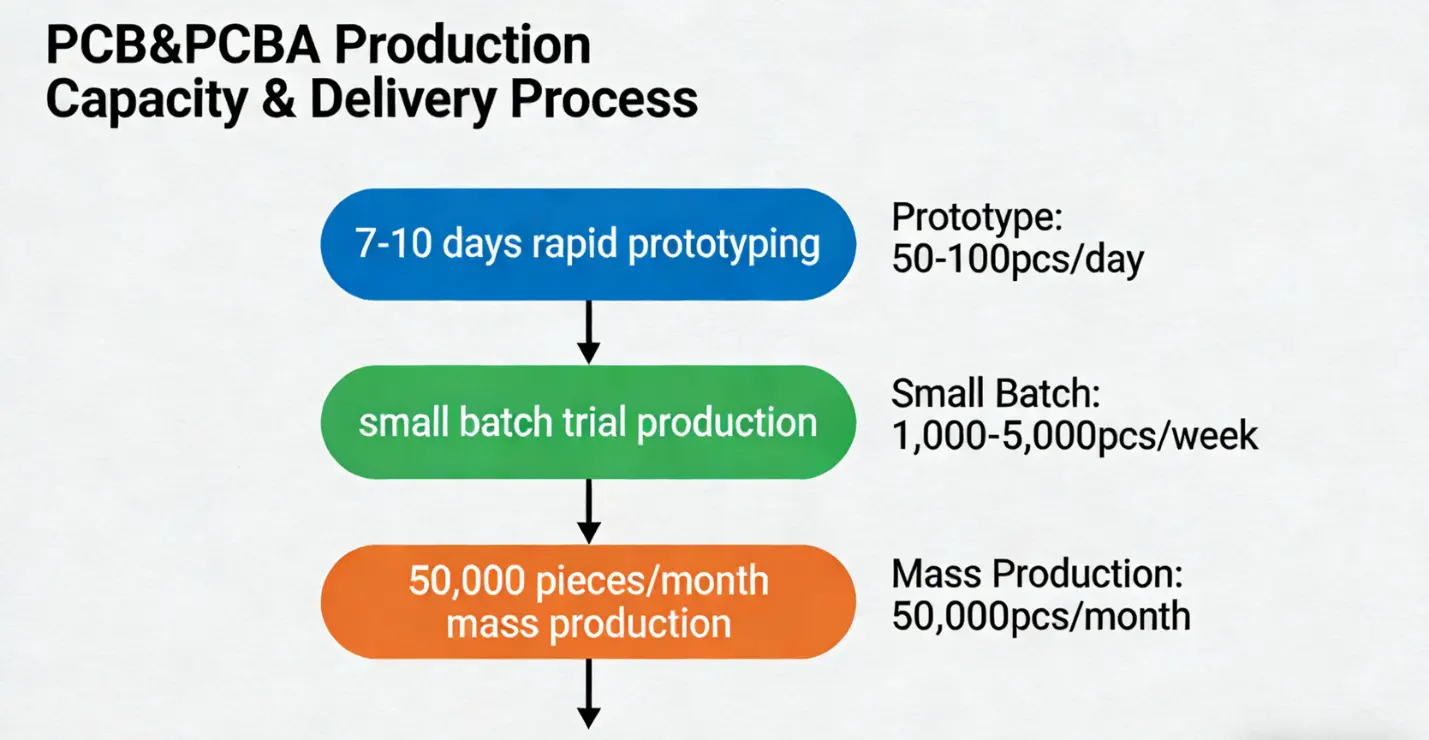
We specialize in complex, low-volume productions typical of Class II and Class III medical devices. We support rapid prototyping (7-10 working days for sample delivery) and scalable mass production, with a monthly capacity of up to 50,000 units for Class III device PCBs. Even for minimum orders as low as 10 units, we maintain strict process control without compromising quality. For emergency orders, we offer a 48-hour rapid response mechanism to meet urgent project needs.
Equipped with high-speed, vision-guided placement machines (±15 µm repeatability) and nitrogen-enriched reflow ovens, we excel at handling ultra-fine-pitch components—down to 01005 (the smallest standard passive component in the industry, representing cutting-edge integration capabilities) and 0.3 mm CSP (Chip Scale Package), which are widely used in miniaturized therapeutic modules. In mass production, the average placement yield for these precision components reaches 99.97%.

For devices exposed to bodily fluids or harsh environments, we apply medical-grade conformal coatings and selective potting, with clear differentiation of applicable scenarios:
· Acrylic coating: Cost-effective, suitable for non-implantable devices requiring basic corrosion resistance;
· Urethane coating: Offers excellent moisture and chemical resistance, ideal for home therapy equipment;
· Parylene coating: Biocompatible and hermetically sealed, exclusively designed for implantable devices to ensure long-term insulation and resistance to bodily fluid corrosion.

While final assembly is completed in partner Class 7 cleanrooms, our pre-assembly workflows strictly follow ISO 14644-1 Class 8 standards—including low-particulate handling, ESD-safe work zones, and ionized workstations. This minimizes contamination risks, laying a solid foundation for subsequent sterilization and assembly processes.
In therapeutic applications, a single solder void or misaligned component can have fatal consequences. Our quality system is built on proactive risk mitigation, with data-driven guarantees:
We hold ISO 13485:2016 certification (the global gold standard for medical device quality management) and comply with IEC 60601-1 (safety requirements for medical electrical equipment), RoHS, and REACH. Additionally, we have obtained FDA registration (for the U.S. market), CE MDR certification (for the EU market), and NMPA filing (for the Chinese market), enabling seamless market access for our clients’ products worldwide. All materials are fully documented with Certificates of Conformity (CoC) and RoHS declarations, ensuring full compliance throughout the supply chain.
· SPI (Solder Paste Inspection): 3D laser scanning ensures optimal paste volume and alignment before component placement, with a paste volume accuracy of ±5%.
· Automated Optical Inspection (AOI): Post-reflow inspection detects tombstoning, bridging, and component skew, achieving a defect capture rate of over 99.5%.
· 2D/3D X-Ray (AXI): Critical for validating BGA, QFN, and hidden joints, ensuring solder voiding
· Flying Probe & ICT/FCT Testing: Functional testing simulates real-world operating conditions (e.g., temperature fluctuations, voltage variations), validating power sequencing, signal integrity, and fail-safe mechanisms. The pass rate for functional testing in mass production exceeds 99.9%.

Every board is assigned a unique serial number. From raw laminate lot numbers, component date codes, and reflow profiles to test results, full genealogy is stored in our Manufacturing Execution System (MES). This enables rapid root-cause analysis and recall readiness. Over the past 5 years, our Class III device PCBA delivery has maintained a defect rate (ppm) of
PCBMO’s therapeutic PCBA solutions are tailored to high-stakes medical segments, addressing core pain points of each application:
Neurostimulators, pacemakers, and cochlear implants—requiring hermetic sealing compatibility and ultra-low leakage currents (<0.03%.
Insulin pumps, PCA (Patient-Controlled Analgesia) units, and smart inhalers—demanding precise motor control (±0.01 mm positioning accuracy) and battery safety (overcharge/overdischarge protection with a response time of
TENS units, wearable defibrillators, and transdermal patches—integrating flexible circuits and biocompatible materials. Our DFM (Design for Manufacturing) team specializes in optimizing flexible circuit durability, ensuring over 10,000 bending cycles without performance degradation.
CPAP machines, dialysis controllers, and phototherapy systems—needing robust EMI shielding (meeting CISPR 22 Class B standards) and continuous-duty reliability. Our PCBA solutions support 24/7 uninterrupted operation, with a mean time between failures (MTBF) exceeding 100,000 hours.
We are more than a contract manufacturer—we are an extension of your R&D and quality teams, addressing core pain points throughout the cooperation lifecycle:
Our DFM/DFA (Design for Assembly) reviews focus on critical medical device requirements:
· Sterilization compatibility: Validating performance stability after EtO (Ethylene Oxide, a common low-temperature sterilization method), gamma, or e-beam sterilization;
· Thermal management: Optimizing heat dissipation in sealed enclosures to prevent component failure due to overheating;
· Design-for-testability: Embedding test points to reduce field failure rates by 30% on average.
· Dedicated medical production line: Physically segregated from consumer electronics workflows, eliminating cross-contamination risks and ensuring strict adherence to medical-grade processes;
· Patent-backed technology: Holding 8 core patents, including a self-developed "nano-parylene composite coating process" that enhances bodily fluid corrosion resistance by 30% compared to standard coatings;
· Superior cleanroom control: Our pre-assembly Class 8 cleanroom maintains a particulate count 50% lower than industry standards, reducing contamination-related defects by 90%.
· Supplier management: Only collaborating with Tier 1 component suppliers (e.g., Texas Instruments, STMicroelectronics) with ISO 13485 certification; establishing a dual-supplier system for key components to avoid supply chain disruptions;
· Risk mitigation: Formulating contingency plans for geopolitical impacts and component shortages, including strategic inventory reserves for critical materials;
· After-sales guarantee: Offering a 3-year warranty for Class III device PCBs (industry average: 1-2 years); providing 24-hour technical support with on-site troubleshooting within 72 hours for global clients;
· Transparent cooperation process: Prototyping → DFM optimization → small-batch trial production (100-500 units) → mass production, with regular progress reports and joint quality reviews at each stage.
Ready to turn your therapeutic device design into a reliable, compliant product? Take the following actions to start our cooperation:
· Apply for afree DFM assessment to optimize your design for sterilization, thermal management, and cost-efficiency;
· Schedule a virtual or on-site tour of our Class 8 cleanroom and dedicated medical production line;
· Contact our medical device specialists at PCB@pcbmo.com o for a customized solution quote.
Substrate Material Parameters
Substrate material parameters are the core guarantee for the stability of therapeutic device PCBA, with the following key requirements:
• Base Material Type: Core substrates include FR-4 (High Tg ≥ 170°C), high-frequency Rogers laminates, and flexible Polyimide (PI). High Tg substrates are suitable for high-temperature sterilization scenarios, while flexible PI is adapted for wearable therapeutic devices.
• Glass Transition Temperature (Tg): The specification range is 170°C to 220°C, which can effectively resist deformation during reflow soldering and autoclave sterilization.
• Coefficient of Thermal Expansion (CTE): 12–18 ppm/°C for X/Y axis and 60–80 ppm/°C for Z axis, minimizing warpage caused by thermal stress and adapting to PCBAs integrated with precision sensors.
• Dielectric Constant (Dk): The dielectric constant is 3.5–4.5 at 1 GHz, ensuring signal integrity of control circuits in therapeutic devices.
• Chemical Tolerance: Resistant to disinfectants such as ethanol and hydrogen peroxide, meeting the repeated sterilization requirements of clinical therapeutic equipment.
Circuit Design & Layout Parameters
Circuit design and layout parameters directly affect the integration and performance of PCBA, with the following critical requirements:
• Minimum Trace Width/Space: Ranging from 30/30 μm (1.2/1.2 mil) to 50/50 μm (2/2 mil), the compact spacing design is suitable for miniaturized PCBAs of implantable therapeutic devices.
• Base Copper Thickness: Optional 1 oz (35 μm), 2 oz (70 μm), 4 oz (140 μm). Among them, 2–4 oz copper is applicable for PCBAs of high-current therapeutic devices such as defibrillators.
• Via Diameter (Drilled/PTH): Micro-via specifications are 50–100 μm, and through-hole vias are 0.3–0.8 mm. Blind/buried via design can meet the layout needs of high-density multi-layer PCBAs.
• Characteristic Impedance Tolerance: ±5% to ±10%, a mandatory requirement for PCBAs of RF therapeutic devices such as microwave ablation equipment to ensure stable signal transmission.
• SMT Pad Size Tolerance: Controlled within ±10 μm to ensure solder joint stability of tiny components such as 0201 and 01005.
Manufacturing Process Parameters
Manufacturing process parameters determine the reliability of PCBA, with core requirements for each link as follows:
• Surface Coating Type: Optional Electroless Nickel Immersion Gold (ENIG), Organic Solderability Preservative (OSP), and Immersion Silver. ENIG is preferred for long-term reliability, while OSP is suitable for cost-sensitive non-sterile devices.
• Pick-and-Place Accuracy: The placement accuracy is ±0.05 mm for 0201 components and ±0.1 mm for packaged components such as QFP/BGA. High-precision placement adapts to the BGA/CSP packaging needs of therapeutic device control modules.
• Reflow Temperature Profile: Peak temperature 245°C ±5°C, Time Above Liquidus (TAL) 60–90 seconds, adopting lead-free process compliant with RoHS 2.0 standard.
• Wave Temperature & Speed: Wave temperature 260°C ±5°C, conveyor speed 0.8–1.2 m/min, applicable for soldering through-hole components of external therapeutic device PCBAs.
• Coating Material & Thickness: Optional Parylene C, Silicone, Acrylic, with thickness controlled at 20–50 μm. Among them, Parylene C is suitable for biocompatible implantable therapeutic device PCBAs.
Quality & Reliability Test Parameters
Quality and reliability test parameters ensure that PCBA meets clinical use standards, with specific requirements as follows:
• Solder Joint Shear Strength: ≥ 5 N per 0402 component, ≥ 15 N per QFP pin, complying with IPC-A-610 Class 3 standards.
• Thermal Cycling Conditions: 1000 cycles at -40°C to 85°C, 30 minutes dwell time per cycle, verifying the temperature resistance and durability of portable therapeutic device PCBAs.
• Damp Heat Test: 1000 hours at 85°C/85% relative humidity, preventing corrosion caused by moisture in clinical environments.
• Insulation Resistance: ≥ 10^9 Ω at 500 VDC, meeting the electrical safety requirements of IEC 60601-1.
• Hipot Test Voltage: 1500 VAC for 1 minute, a mandatory test requirement for PCBAs of therapeutic devices powered by mains electricity.
Compliance & Regulatory Parameters
Compliance and regulatory parameters ensure that PCBA meets global medical device market access standards, with specific requirements as follows:
• Quality System Standard: Compliant with ISO 13485:2016, a mandatory system requirement for medical device PCBA manufacturing.
• Medical Device Safety Standard: Compliant with IEC 60601-1 (Edition 3.2), covering basic safety and essential performance requirements.
• U.S. Regulatory Compliance: Compliant with FDA 21 CFR Part 820 (QSR), a necessary condition for entering the U.S. market.
• RoHS/REACH Compliance: Meeting RoHS 2.0 (2011/65/EU) and REACH regulations (SVHC list), restricting hazardous substances in PCBA materials.
• Biocompatibility Standard: Compliant with ISO 10993, applicable for implantable or skin-contacting therapeutic device PCBAs.